Everything The Biotech Ecosystem Should Know About Antibody Discovery Services


THE EVER-EVOLVING FIELD OF ANTIBODIES
Fit-for-Purpose Approach to Antibody Discovery
The antibody discovery field is growing rapidly, driven by platform innovations, increased understanding of immunology, and the application of sequencing and data science to uncover broader insights more efficiently. As technology in antibody discovery continues to improve, the possibilities and applications for antibody-based therapeutics are unlocking new white space opportunities such as targeting challenging and previously undruggable targets and novel often multimodal therapeutic mechanisms of action.
Every day our partners bring us new and exciting therapeutic hypotheses each posing unique challenges and target product profile requirements. We believe there is no one-size-fits-all approach to enable success across these variety of exciting programs. Within the antibody discovery field there are a breadth of discovery approaches and technologies to select from each with their own merits and tradeoffs. That is why, at Alloy, we have a broad range of antibody discovery platforms and take a fit-for-purpose approach to designing each antibody discovery campaign catered to the desired therapeutic profile in mind from the very beginning.
The following is a general overview of the antibody discovery process, with commentary around how one may begin to think about the options along the way that may best fit their needs.

>170
60+

11

DISCOVERY APPROACHES
How Does Antibody Drug Discovery Work?
Generating Diversity
The human adaptive immune system is incredibly sophisticated, capable of producing a vast array of antibodies through mechanisms such as VDJ recombination and somatic hypermutation. VDJ recombination involves the random rearrangement of gene segments encoding the variable regions of antibodies, leading to the creation of a diverse repertoire of antibody sequences. Additionally, during an immune response, B cells undergo somatic hypermutation, introducing random mutations in the antibody variable regions, further increasing diversity. This diversity allows the immune system to effectively recognize and neutralize a wide range of antigens.
In antibody drug discovery, mimicking this natural process is crucial. Techniques such as immunization of animals such as transgenic mice or in vitro selection methods like phage and yeast display are employed to generate a large pool of diverse antibody candidates representing various sequences and specificities. At Alloy a majority of our diversity generation begins with our our proprietary suite of transgenic mice expressing full human antibody diversity (ATX-Gx) while we also have comprehensive expertise in both phage and yeast display. This initial step lays the foundation for subsequent screening and selection processes.
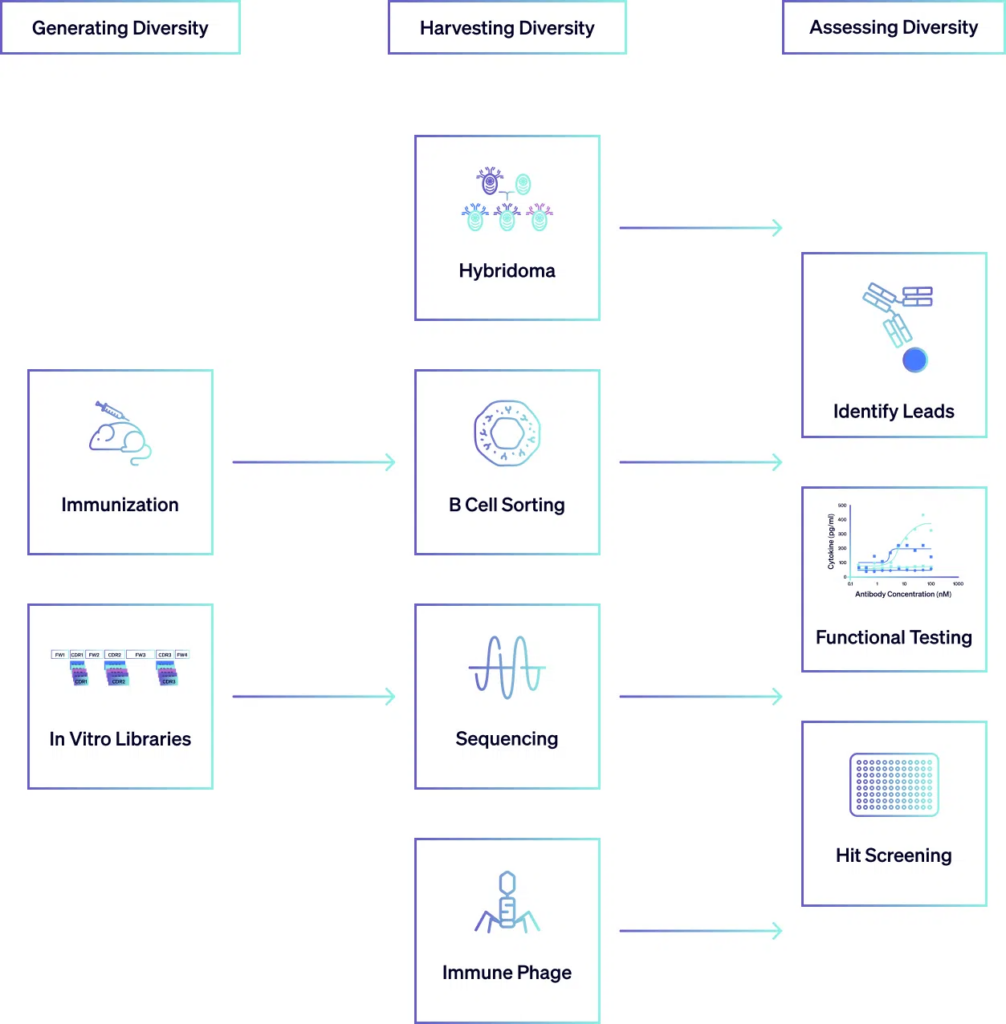
Harvesting Diversity
Once a diverse pool of antibodies is generated, the next step is to isolate those antibodies with desired properties, such as high specificity and affinity for the target antigen. This is achieved through a variety of techniques tailored to the specific characteristics of the antibodies and the target antigen. For example, ELISA and FACS are commonly used to screen large antibody libraries for binding to the target antigen. Additionally, bulk NGS sequencing bulk can also be employed to analyze the diversity of the antibody repertoire and identify sequences that are enriched for binding to the target antigen. In phage display libraries, biopanning involves multiple rounds of selection to enrich for phages displaying antibodies with desired binding properties. Through these selection processes, antibodies with the most promising characteristics are identified and isolated for further assessment.
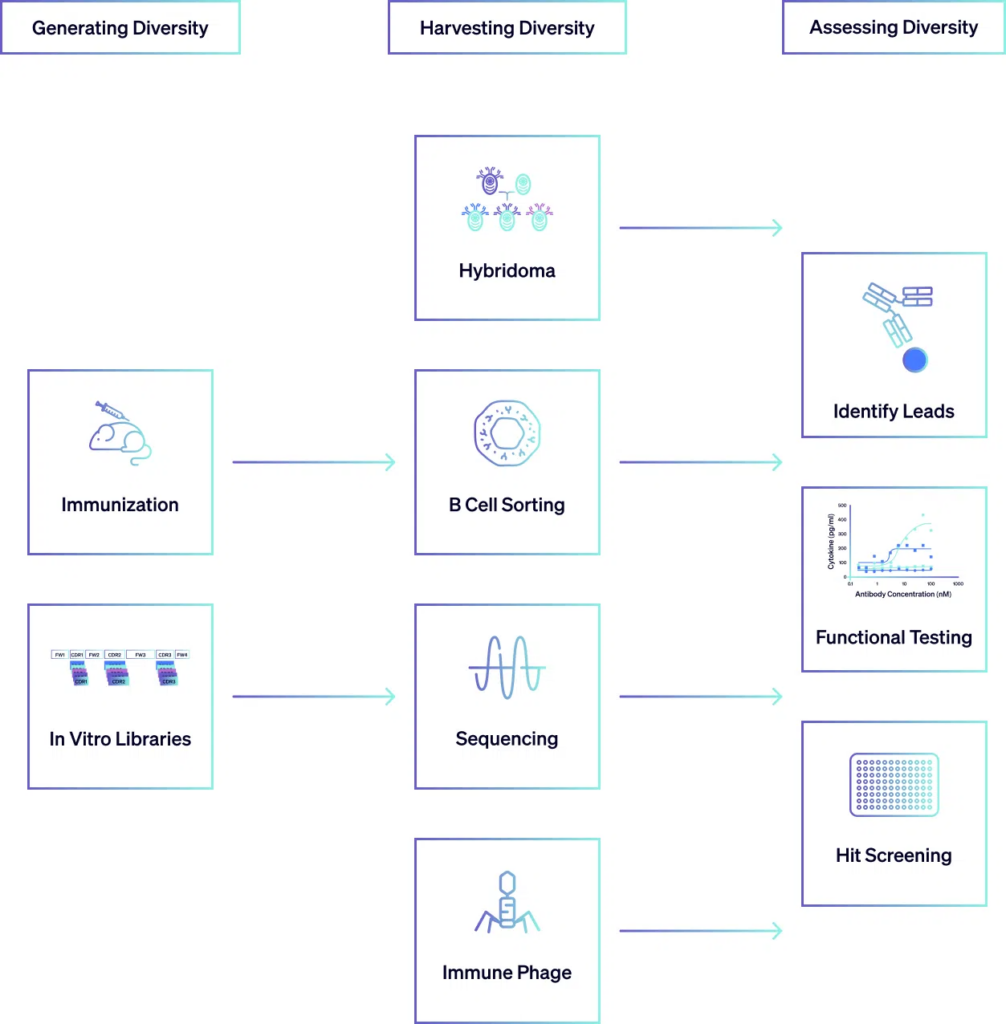
Assessing Diversity
The antibodies selected from the screening process undergo comprehensive evaluation to assess their efficacy and suitability for therapeutic use. Following initial screening, antibodies undergo rigorous analysis of their binding properties including assessing parameters such as binding kinetics, epitope specificity, and cross-reactivity with related antigens. Functional assays are conducted to determine whether the antibodies can effectively modulate the intended disease target and mode-of-action. Cell-based assays and in vivo animal models are commonly used to assess antibody function in relevant biological systems. Additionally, the selected antibodies are assessed for developability criteria, which encompass factors such as solubility, stability, aggregation propensity, and manufacturability. This extensive assessment ensures that the antibodies meet the necessary criteria paving the way for their development into therapeutic agents.

ANTIBODY DISCOVERY TECHNIQUES
What Are The Techniques Used in Antibody Discovery?
Antibody discovery involves various techniques aimed at identifying and isolating antibodies with desired properties. First, diversity can be generated either through in vivo immunization approaches or synthetic in vitro libraries. For immunizations, animals (such as mice, rabbits, camelids, etc.) are immunized with a target antigen to stimulate an immune response and serum containing antibodies are collected and screened for the desired specificity. In these approaches antibodies can be harvested in a variety of manner such as hybridoma, single b-cell sorting, and bulk NGS sequencing to capture the antibody repertoire. In contrast, in vitro library diversity is generated from the construction of libraries of antibody fragments (such as scFv or Fab) displayed on phage, yeast, or other expression systems which are then screened for antigen binding. In some cases, it may be useful to import sequence information from an immunized animal into an in vitro library to be panned further.
Lastly, antibodies harvested from these processes must be produced to assess their binding, functional, and developability properties. It is crucial to be able to produce high quality antibodies and assay them in a high-throughput manner in order to effectively screen a breadth of leads. At Alloy we have established a variety of high-throughput production and screening methodologies on the Carterra LSA Platform and using high-throughput flow cytometry to rapidly select for the right leads candidates. Additionally, Alloy has the necessary protein engineering capabilities to reformat the desired leads into a variety of modalities for functional testing including bi- and multi-specific antibody formats, chimeric antigen receptors, and more.
What is B cell sorting for Antibody Discovery?
Process: B cells are isolated from single cell suspensions of lymphoid tissue based on their ability to produce antibodies specific to your target of interest. This is often accomplished using FACS with a fluorescently labelled target antigen as the staining reagent, along with other fluorescent antibodies for identifying particular B cell lineages.
Benefits: Once isolated to high purity, target-specific B cells can be processed directly for antibody sequencing, as the sorting has already served as the target-binding screening step in this binder recovery process. This allows for more rapid progression from cell isolation to binder recovery compared to the extended culture times required for hybridoma screening.
Potential drawbacks: The ability to perform B cell sorting is critically dependent on the nature of your target antigen, requiring extensive validation of the proposed sorting process prior to its use for a specific antibody discovery campaign. For example, if your target is a soluble protein or a single pass transmembrane protein that can be expressed as a soluble construct (e.g. Fc-fusion), then you should be able to generate fluorescently labelled antigen as a FACS reagent. However, if your target is more challenging, such as a multi-pass transmembrane receptor, the ability to generate soluble FACS reagents may be limited. Additionally, B cell sorting methodologies often require antibody display on the surface of cells in the form of B cell receptors, meaning that sorting methodologies often focus on memory B cells, unlike hybridomas that can also encompass antibody-secreting plasma cells. Next generation sequencing (NGS) of the antibody repertoire parallel to any discovery approach helps to capture these plasma cells sequences and overcome this limitation. Alloy employs deep repertoire sequencing on all of its discovery campaigns to enrich the output diversity and enable us to access more antibody diversity for testing
What is Hybridoma for Antibody Discovery?
Process: Spleen and/or lymph node cells are fused with myeloma cells to generate immortalized cells, which are then screened for the production of target-binding antibodies. Hybridoma technology is a time-tested approach to binder recovery from immunization campaigns, and while the recovery process may be long and labor-intensive, it can reliably generate a large panel of potential leads from any manner of antibody discovery campaign.
Benefits: By coupling hybridoma generation with cell culture methods such as limited dilution cloning or culture in semi-solid media, the hybridoma process can yield monoclonal antibodies with relative ease. Additionally, as long as the cloning procedure was performed correctly, these antibodies will retain their correct heavy- and light-chain pairing, as well as any post-translational modifications that improve stability and limit aggregation.
Potential drawbacks: The production and screening of hybridomas can be a time-intensive process, especially when tasked with screening hundreds of potential clones for target-binding antibody production. This limits the throughput of the approach leading to fewer antibodies being screened during a typical campaign thus decreasing the probability of finding unique antibodies that fit a strict target product profile.
What is Phage Display for Antibody Discovery?
Process: Libraries of antibodies either synthetically produced or directly recovered from lymphoid organs are displayed on the surface of bacteriophages, usually in either the Fragment antigen-binding (Fab) format or as single-chain fragment variables (scFv). These antibody fragment-displaying phages can then be “panned” against your target of interest to enrich phages that express variable regions with target-binding properties.
Benefits: Phage display allows for enrichment of particular target-binding properties during the panning process—for example, binding to particular epitopes on your target, or cross-reactivity to multiple species. Panning can also be used in a subtractive fashion, such as to enrich for antibodies that bind only one variant of a larger related family of proteins. Additionally, phage libraries can be interrogated multiple times in future panning rounds to optimize recovery of binders. Phage libraries can also be leveraged for in vitro affinity maturation of antibodies, a powerful tool for improving the binding affinities of potential leads.
Potential drawbacks: As phage libraries rely on display of Fab or scFv versions of full-length antibodies, there may be unforeseen developability issues with identified leads when they are reformatted back to full-length antibodies. In addition, as the library generation process typically either involves design of synthetic repertoires and/or randomization of the heavy- and light-chain pairing, there is an increased risk that leads identified through phage panning may lead to less well-behaved full-length antibodies.

TYPES OF ANTIBODY THERAPEUTICS
What Are The Different Types of Therapeutic Antibodies?
Therapeutic antibodies can be derived from various species, each with their advantages and limitations. Murine antibodies originate entirely from mice, exhibiting effectiveness in therapy but often provoking immune reactions in humans, thus limiting their long-term utility. Chimeric antibodies, on the other hand, combine human and mouse elements, featuring human constant regions and mouse variable regions, thereby mitigating immune responses in patients but only to a limited degree and still limiting long-term dosing.
Humanized antibodies predominantly comprise human components with non-human-derived antigen-binding sites, further diminishing immunogenicity risks. Fully human antibodies are exclusively sourced from humans, achieved through techniques like transgenic animals or phage display, minimizing the likelihood of eliciting immune responses in patients. Other host species for antibody discovery are being utilized for immunization and subsequently humanized because of their unique structural properties (including camelids, sharks, chickens, and bovine).
At Alloy we focus our approach on generating fully human antibodies utilizing our best-in-class transgenic murine platform producing the full repertoire of human antibody sequence space. Additionally, Alloy has specialized technologies for bispecific and multispecific antibody generation including out common light chain platform (ATX-CLC) as well as our single domain libraries engineered for stability and resistance to aggregation.
Therapeutic antibodies can also be formatted into a variety of modalities to enable unique mechanisms of action as listed below. Alloy has the protein engineering and high-throughput functional screening capabilities to rapidly produce and test antibodies in a variety of these formats to evaluate lead candidates in the proper modality and disease context:
Monoclonal antibodies (mAbs)
These are antibodies produced by identical immune cells and are engineered to bind to specific targets, such as proteins or receptors, on cells. The main mechanism of action for mAbs is to block the function of these targets, inhibit signaling pathways, or trigger immune-mediated destruction of target cells.
Bispecific antibodies
Bispecific antibodies are designed to bind to two different targets simultaneously. They can redirect immune cells to target cells, such as tumor cells, by binding to both the target cell and an immune cell, such as a T cell or a natural killer (NK) cell. This mechanism enhances the specificity and efficacy of immune-mediated destruction of target cells. Other applications include development of agonistic antibodies activating and inducing receptor signalling.
Antibody-drug conjugates (ADCs)
ADCs combine the targeting ability of antibodies with the cytotoxic potency of small-molecule drugs. The antibody component of an ADC selectively binds to a target antigen on the surface of cancer cells, delivering the attached cytotoxic drug directly to the cancer cells. Upon internalization, the drug is released, leading to cell death.
Fc-engineered antibodies
Antibodies can be modified in their Fc region to enhance or alter their effector functions, such as antibody-dependent cell-mediated cytotoxicity (ADCC) or complement-dependent cytotoxicity (CDC). These modifications can improve the antibody’s ability to recruit immune cells or complement proteins to destroy target cells, or silence the Fc region for applications where effector function is not warranted. Other, mutations can be included to increase half-life.
Single-Domain Antibodies
Small antibody fragments based on naturally occurring single-domain antibodies found in camelids, such as llamas and camels. They offer advantages such as high stability, solubility, and tissue penetration, making them attractive for therapeutic applications. Exert their effects by blocking protein-protein interactions, inhibiting enzymatic activity, or delivering payloads to target cells.
Chimeric antigen receptor (CAR)
CAR are engineered antibodies that incorporate antigen-recognition domains from antibodies along with signaling domains from T cells. These CAR antibodies are used to modify a patient’s own T cells, enabling them to recognize and kill cancer cells expressing specific antigens.
Antibody-based protein degraders (ABD)
ABDs are a class of molecules that utilize antibodies to target disease-causing proteins for degradation. Through the selective binding of antibodies to specific target proteins, ABDs recruit cellular protein degradation machinery, leading to the degradation of the targeted proteins. This approach holds promise for treating diseases by eliminating pathogenic proteins.

ANTIBODY DISCOVERY CHALLENGES
What are the Challenges in Antibody Discovery?
The discovery of therapeutic antibodies encompasses numerous challenges. Initial steps involve target identification, necessitating an understanding of disease biology to pinpoint disease-specific targets. Selecting the right antigen poses difficulties due to factors such as availability and immunogenicity therefore approaches such as DNA and mRNA-LNP immunization are crucial in overcoming these factors.
Generating a diverse pool of antibodies for screening requires alternative approaches like single B-cell sorting, phage display, and NGS therefore having all approaches available enables a bespoke approach based on the specific target product profile criteria. Additionally, screening large antibody libraries for lead candidates is resource-intensive, requiring advanced analytical techniques for accurate characterization thus high-throughput methodologies are helpful in streamlining this process. Ensuring a lead candidate demonstrates the desired efficacy requires testing for functional characteristics in cell-based assays early and in the desired format to ensure the right leads are prioritized for further characterization.
Lastly, a comprehensive developability screening approach is required to ensure lead candidates show all the desirable drug-like properties required to progress into preclinical studies. Overcoming these obstacles demands interdisciplinary collaboration and innovative technologies. Despite the challenges, advancements in discovery methodologies continue to propel the development of novel antibody-based therapeutics across various diseases.

DO YOU HAVE A QUESTION FOR US?
FAQ
What is Antibody Discovery?
Antibody discovery is the process of identifying and developing antibodies that can specifically target and bind to a particular antigen. This process typically involves screening large numbers of antibodies to find ones with the desired therapeutic properties, such as high affinity, target specificity, developability, etc. Once potential antibodies are identified, they undergo further optimization and testing to ensure their efficacy and safety for use in various applications. Antibody discovery methods have evolved significantly, incorporating techniques such as B-cell sorting, phage display, hybridoma technology, and next-generation sequencing, allowing for more efficient and precise antibody development.
Why is Antibody Discovery Important?
Antibodies are an important contributor to modern medicines with nearly two hundred approved antibody-based therapies and hundreds more in clinical development, spanning a wide range of diseases including cancer, autoimmune, cardiometabolic, CNS conditions, and more. Their ability to bind target antigens with high affinities and specificities, combined with their versatility of function, make them a fundamental building block for a variety of more complex therapeutic modalities, including antibody-drug conjugates, bispecific antibodies, and immune checkpoint inhibitors, revolutionizing the landscape of medical treatments. The success of antibody-based therapies underscores the significance of antibody discovery in driving innovation and improving patient care across diverse healthcare fields.
What Is An Antibody Platform?
An antibody platform refers to a comprehensive approach or technology that facilitates the discovery, development, and optimization of antibodies for therapeutic or diagnostic purposes. These platforms typically integrate various techniques, such as high-throughput screening, computational modeling, and molecular engineering, to efficiently generate antibodies with desired properties, including high affinity, specificity, and stability. Antibody platforms play a crucial role in accelerating the drug discovery process by streamlining antibody production, characterization, and optimization, ultimately leading to the creation of more effective and targeted treatments for a wide range of diseases.
What Is The Process of Antibody Development?
The process of antibody development typically involves identifying a lead candidate through discovery, optimization, and screening, before proceeding to preclinical testing prior to advancing to clinical trials. Antibodies that emerge from the discovery process are screened until a lead candidate is identified that meets all aspects of the desired target product profile, including but not limited to the right affinity, functional criteria, and developability properties. Next, preclinical testing entails evaluating the safety, efficacy, and pharmacokinetics of the developed antibodies in animal models to gather essential data for regulatory submissions. Once the preclinical stage is successfully completed, the antibodies advance to clinical trials, where they undergo rigorous testing in human subjects to assess safety and efficacy. These trials are typically conducted in three phases, each with increasing numbers of participants and stringent regulatory oversight. Finally, if the antibodies demonstrate safety and efficacy in clinical trials, they may receive regulatory approval for use in patients, marking the culmination of the antibody development process.
What is the Best Method for Antibody Discovery?
Considering that each unique approach has specific strengths and weaknesses, how do you choose the approach that is best for your antibody discovery program? As mentioned above, the nature of your target antigen is often the defining factor. If you have a well-behaved antigen that can function as a soluble construct, single B cell sorting is a powerful approach. However, if your target antigen is more challenging, hybridoma or immune phage may be the optimal approach. Ultimately, it is important to recognize that no single approach is necessarily best for identifying the best diversity of potential leads which is why a fit-for-purpose approach towards antibody discovery focusing on the target product profile in the design of a campaign is important towards increasing the probability of a successful therapeutic outcome.
ABOUT ALLOY
Alloy Theraputics
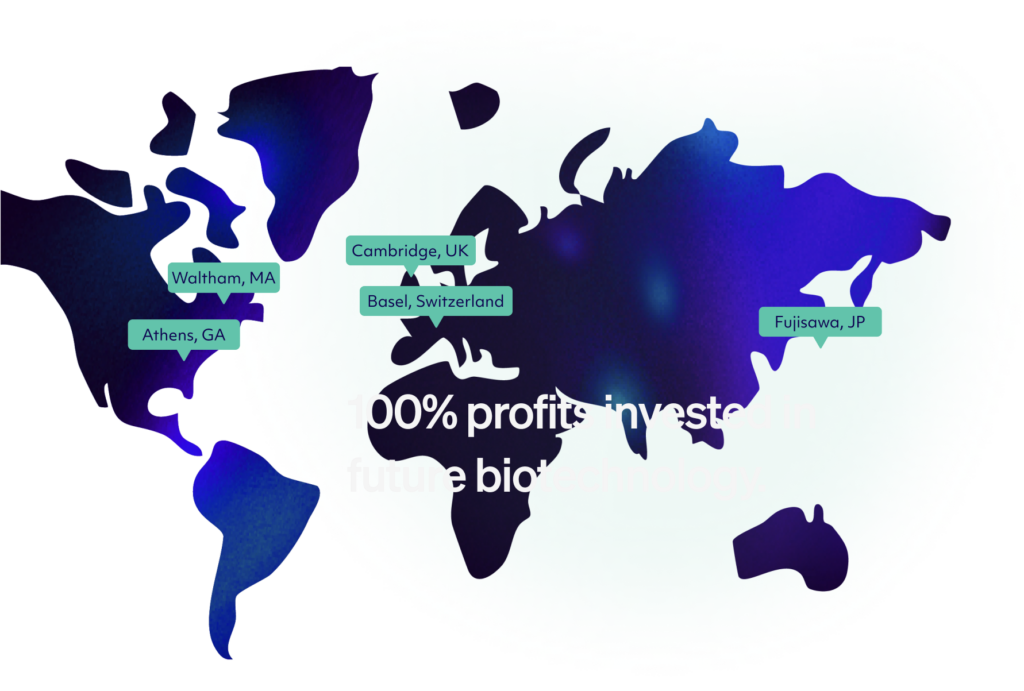
Alloy Therapeutic is a trusted partner in fit-for-purpose therapeutic antibody discovery, with over 150 partners to date and counting. Alloy is led by a scientific team that has put hundreds of antibodies into the clinic that is enabled by an ever-growing stable of antibody discovery platforms that blend the best of in vivo and in vitro wet-lab capabilities with AI/ML. Founded in 2017 and privately funded by visionary investors, Alloy is headquartered in Boston, MA, with locations in Athens, GA and internationally in Basel, CH and Cambridge, UK. We also reinvest 100% of our profits back into our technologies and people to make sure we are always staying ahead of the curve.

Antibody Platform
Pioneering Transgenic Mouse Technology
The star and the start of our workflow is the industry-trusted transgenic ATX-GX Mouse Suite for fully human antibody discovery. The workhorse mouse, ATX-GK, generates a robust immune response and a proven source of diverse sets of antibodies towards a variety of targets while our Common Light Chain Mouse Suite, ATX-CLC, streamlines the creation of bispecific and multi-specific antibodies.
ALLOY ANTIBODY DISCOVERY SERVICES
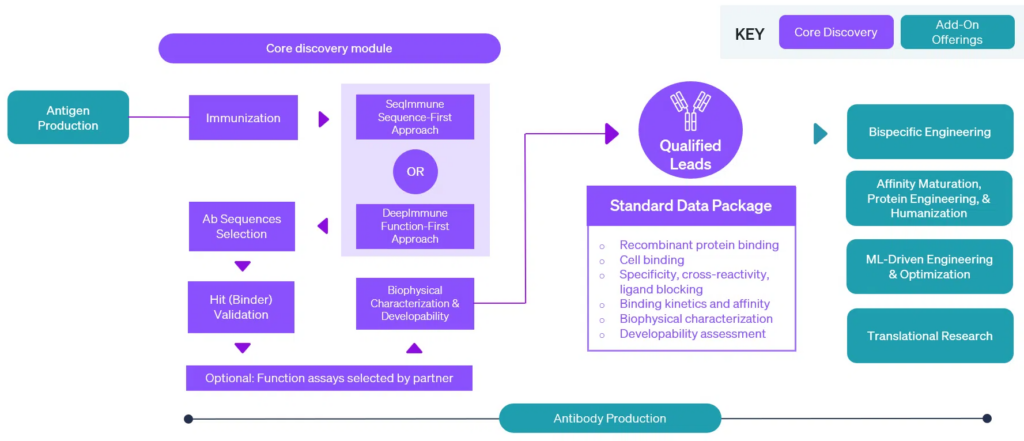
A Fit-for-Purpose Approach to Delivering Lead Antibody Candidates
From this start, we have built an industry-trusted end-to-end integrated workflow that starting from custom antigen production (including mRNA-LNP and DNA vectors) and immunizing the mouse using the proven protocols, to delivering you the best leads, be they simple monoclonal antibodies, or complex bi- and multi-specifics.

Start your next discovery campaign with us
